Efficient digital corridors for patients with rare diseases


Boehringer Ingelheim is one of the world’s largest pharmaceutical companies, and the largest private one.
The company’s key areas of interest are respiratory diseases, metabolism, immunology, oncology, and diseases of the central nervous system.
In the last year, we worked together with our partners from BI from different departments in their organization:
- Boehringer Ingelheim Regional Center Vienna
- Boehringer Ingelheim Romania
- Open-Source Imaging Consortium for ILDs
Key Numbers
A couple of key numbers that turn this implementation into success:
- 65% decrease in the time it takes for patients to be diagnosed from their first HR-CT investigation: from 12 months to 3-4 months.
- Medicai organized 40 diagnostic boards (multi-disciplinary team), several times per month, online, remotely and efficiently.
- Doubled the number of patients that benefited from treatment for IPF in 2020, compared to 2019.
- 4 centers of excellence in Romania in IPF/ILDs were integrated with Medicai.
- Our project represented the Central Eastern European Region in the Global GTM Awards competition.
- 100+ specialists (pulmonologists and radiologists) and 20+ IPF/ILDs experts can offer advice to 400 + pulmonologists in Romania.
- We are planning to expand to other countries in the region in 2022.
Challenges
Boehringer Ingelheim focuses on therapeutics and treatment for patients suffering from ILDs, specifically IPF.
The patients’ journey suffering from ILDs / IPF can be structured into steps they take through their patient journey.
Patients’ journey suffering from rare diseases like ILDs/IPF can take several months, if not years from the first suspicion to diagnosis and treatment. Unfortunately, for some patients, this journey takes too long.
We analyzed each step of this journey in order to understand in detail the challenges patients are facing:
Step I
Suspicion to diagnosis (3-6 months)
Delays happen because, usually, there are no experts accessible to the patient and the patient does not know where to go. Navigating the system can take months and, in some cases, even years.
The patient goes to a local specialist, that does the best he can to help the patient and usually tries to determine a differential diagnosis, which takes a couple of months and multiple investigations.
At some point, the doctor contacts experts from the centers of excellence and goes to the next step in the patient journey, the diagnosis.
Step II
Diagnosis (12 months)
Diagnosing rare conditions like IPF/ILDs needs multiple evaluations from multiple doctors. Through the process, the expert doctors require more information from the patients. Sometimes, follow-up medical imaging is required (to analyze the growth etc). The diagnosis process is also bureaucratic (doctors need to gather information, create documents, etc) and, according to studies, it takes 12+ months after a first HR-CT is done.
Step III
Treatment
After a diagnosis is determined, the patient receives a treatment plan. Ideally, a multi-disciplinary team looks over the patient and ensures the patient adheres to the treatment plan.
Summary
The goals of this project were:
- increase the number of IPF patients that are diagnosed and placed under treatment
- reduce the time it takes for them to be diagnosed
Together with our partners from BI, we discovered 2 main challenges that needed to be addressed:
- access to the right expertise as fast as possible
- improving the time it takes to diagnose the patients
Our approach
Context
In 2020, during the COVID pandemic, together with our partners from BI we realized that patients suffering from rare conditions such as IPF are greatly affected by the pandemic.
On one side, they postponed visiting local specialists because they saw it as a health risk. On the other side, the experts from the centers of excellence postponed organizing diagnostic commissions because their workflows changed a lot to improve social distancing in the hospitals.
We realized technology can play a major role and improve the patient journey for rare diseases, such as IPF, even during challenging times like the COVID pandemic.
Improved diagnosis
We started improvements on the patient journey, from suspicion to diagnosis and then treatment. We decided to focus on the diagnosis at first.
For IPF/ILDs, the diagnosis is done in a multi-disciplinary commission, that used to take place offline, in the centers of excellence. Before the commission, when a patient case was discussed and diagnosed, the patient would physically come to the hospital with their patient data (medical imaging, other investigations like DLCO, spirometry) on paper, CDs.
Integrations
Medicai integrated with 4 centers of excellence in Romania and made it very easy for experts working in the centers to access patient data in the hospital, as well as remotely, from home, for example. This has been extremely valuable, especially during the COVID pandemic.
With Medicai, all the patient data is available in the cloud. We integrate with the local hospital infrastructure and the medical imaging is automatically available on the platform. Thanks to our cloud platform, access to patient information is available from within the hospital and remotely, from home.
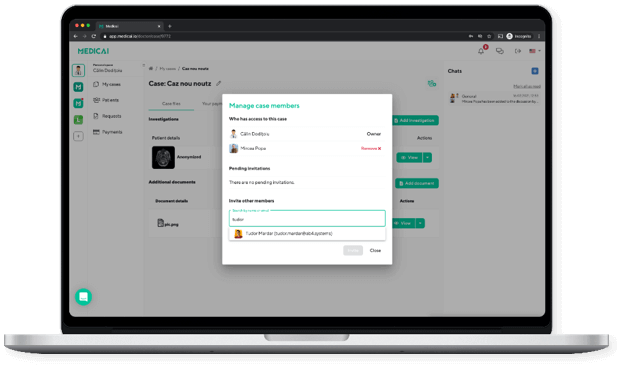
Doctors are in contact with other doctors from the multi-disciplinary team as well as with the patient. The doctors can ask the patients to make additional investigations.
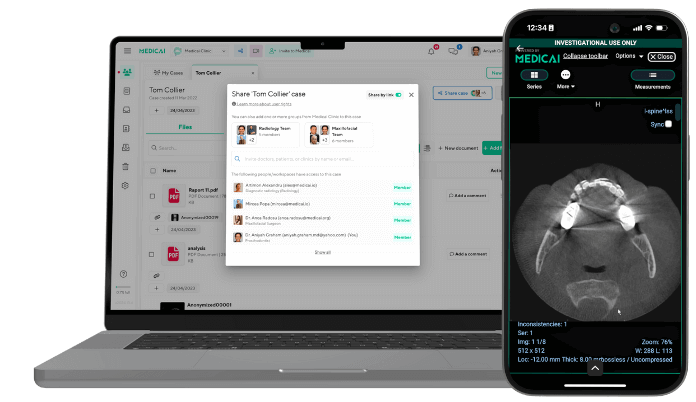
Medicai enables diagnostic boards with audio/video calling and medical information sharing, all within the platform. We also enable digital signing in the platform for diagnostic forms, all in a secure way (HIPAA and GDPR Compliant).
During the past year, 40 diagnostic boards (multi-disciplinary team) were organized several times per month, online, remotely, and efficiently.
With Medicai, we have reduced the time for diagnosis for IPF patients to only 3-4 months from at least 12 months (65% decrease).
Improved suspicion to diagnosis journey
The next step we focused on was the journey of the patient from suspicion to diagnosis.
With Medicai, the patient and the local specialist can organize the medical data efficiently and, thanks to our cloud infrastructure, can access it from anywhere and share it easily.
A panel of experts for specific chronic or rare conditions is one click away from a local specialist or the patient. Collaboration with experts is done efficiently and in a secure, HIPPA and GDPR-compliant way.
This way, the time from suspicion to diagnosis is shortened greatly, sometimes to under a month.
We had patients that were diagnosed remotely, in record time. They did all the investigations locally, with the help of their local pulmonologist, who coordinated with the experts from the centers of excellence.
Improved monitoring during treatment
The next natural step in our efforts to improve the patient journey is to focus on treatment. The treatment for rare diseases can be long and doctors need to make sure patients stick to the treatment.
With Medicai, remote monitoring of patients under treatment is easily done. The doctors can easily communicate with the patient remotely and can ask them to do follow-up investigations in their proximity, without having them come over to the clinic.
Next steps
Thanks to the incredible results we have seen in 2021, a remarkable 65% decrease in the time it takes patients with IPF/ILDs to be diagnosed, we are expanding our partnership with BI.
With Medicai`s technology, we created efficient digital corridors for patients with rare diseases, such as IPF , by drastically reducing the time from suspicion to diagnosis and treatment.
Besides IPF/ILDs, our approach can be replicated in other rare conditions. In 2022, we’re partnering with Boehringer Ingelheim to improve diagnosis for generalized pustular psoriasis (GPP) too.
Related Articles



Lets get in touch!
Learn more about how Medicai can help you strengthen your practice and improve your patients’ experience. Ready to start your Journey?
Book A Free Demo