AI in Telemedicine: How It Is Used in Virtual Care, Remote Monitoring, and Teleradiology

Artificial intelligence in telemedicine is the application of machine learning, natural language processing, and computer vision to support the remote delivery of clinical care. The scope is wider than the chatbot symptom checker that most general healthcare articles focus on. AI in telemedicine spans real-time clinical decision support during video consultations, AI-driven triage and symptom assessment before the visit, remote patient monitoring with AI analytics, ambient AI scribes that handle clinical documentation, and AI-assisted interpretation of medical imaging acquired or shared in telemedicine workflows. Each of these categories addresses a different part of the remote care workflow, and each has a different maturity, regulatory status, and clinical evidence base.
This guide covers the full picture: how AI is used across the five telemedicine application categories, what measurable performance the available tools have demonstrated in clinical validation studies, where the limitations and regulatory boundaries sit today, and how the different application types compare for healthcare organizations evaluating where to invest. The imaging dimension — how AI changes what telemedicine can do when medical images are involved in the consultation — receives particular attention, because it is the dimension most generalist coverage of AI in telemedicine omits and the dimension where the technology infrastructure requirement is most specific. For the broader regulatory and definitional distinction between telehealth and telemedicine, see the telehealth vs telemedicine guide.
How AI Is Used in Telemedicine: Five Application Categories
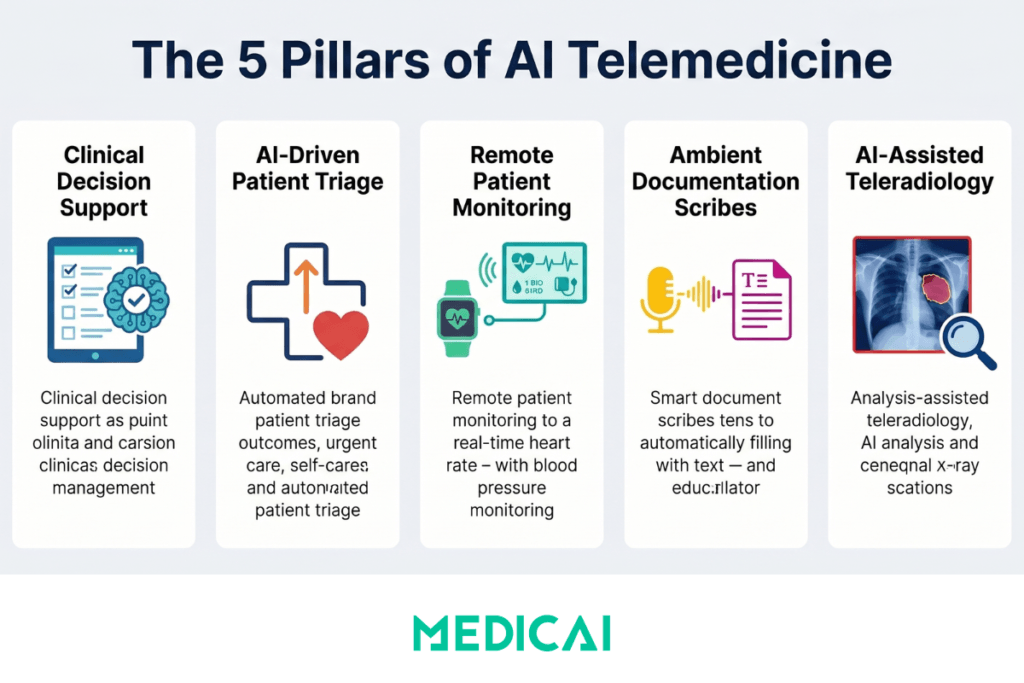
1. Virtual Visit Support and Clinical Decision Support
AI-powered clinical decision support layers operate alongside the live video consultation between the provider and patient, surfacing relevant clinical information when needed. The AI may flag drug interactions when the provider prescribes a new medication during the visit, suggest differential diagnoses based on the documented symptoms, retrieve clinical guidelines applicable to the working diagnosis, or check the visit against the patient’s medical history for missed considerations. The clinical decision support function is not new — EHR-integrated decision support has existed for two decades — but the AI enhancement makes the support more contextual, more responsive to the specific clinical situation, and less reliant on the provider remembering to query the system.
Examples include Epic’s AI-enhanced clinical decision support integrated into the EHR-native telehealth workflow, Cerner’s similar capabilities, telemedicine platforms with native AI overlays such as Amwell’s Converge platform, and the emerging category of LLM-powered clinical assistants used by providers during visits as a real-time reasoning partner. The clinical maturity of decision-support AI is high — the underlying decision logic has been validated for years — but the AI enhancement layer is in active expansion as large language models mature for clinical use.
2. AI-Driven Triage and Symptom Assessment
Pre-visit AI screening tools collect symptom information from patients before they speak with a clinician, apply clinical decision algorithms to the reported symptoms, and either route the patient to the appropriate care setting or schedule the appropriate telemedicine encounter. The clinical goal is operational: directing acute care needs to urgent care or the emergency department, routine concerns to primary care, and specialist needs to specialist consultation, without consuming clinician time on the triage decision itself.
Named products in this category include Buoy Health, K Health, Ada Health, and Babylon’s clinical triage tools. The clinical reality is more nuanced than the marketing suggests. A 2024 BMJ analysis of leading AI symptom checkers found safety advice accuracy ranging from 33% to 79% across the products evaluated, with significant variability in recommendations for identical symptom presentations entered into different tools. The practical implication is that AI symptom triage is clinically useful as an augmentation layer — particularly for reducing low-acuity demand on clinician time — but cannot replace clinician triage judgment for safety-critical decisions where the cost of an incorrect routing is high.
3. Remote Patient Monitoring with AI Analytics
Remote patient monitoring (RPM) generates continuous physiological data from wearable devices, home monitoring equipment, and connected medical devices — a volume that is impossible for clinicians to review manually. AI analytics processes the continuous data stream to detect clinically significant patterns and alert clinicians only when intervention is warranted, transforming a data-volume problem into an actionable clinical workflow.
The most clinically validated application is cardiac arrhythmia detection from continuous ECG monitoring. Apple Watch’s AFib detection feature received FDA clearance based on the Apple Heart Study published in the New England Journal of Medicine, which enrolled over 419,000 participants and demonstrated that the AI-based irregular pulse notification successfully identified atrial fibrillation with a positive predictive value of 84% in the subgroup that completed confirmatory ECG monitoring. Similar AI-enhanced monitoring applications include Dexcom’s continuous glucose monitoring with AI pattern analysis, and AI-enhanced pulmonary function monitoring for chronic obstructive pulmonary disease and asthma management.
CMS Remote Physiologic Monitoring codes (CPT 99453 for initial setup, 99454 for device supply, 99457 for the first 20 minutes of monitoring services, 99458 for each additional 20 minutes) reimburse the monitoring service. The AI analytics layer adds clinical value but does not change the billing structure — payment is tied to the monitoring service and the clinician time, not to the AI layer that makes the monitoring service practical at scale.
4. Ambient AI Scribes and Clinical Documentation
Ambient AI listens to the telemedicine consultation, transcribes the conversation, structures it into clinical documentation, and populates the EHR encounter note. The clinical motivation is direct: documentation burden is consistently identified as the primary contributor to physician burnout across specialties. A clinician who spends 90 minutes after each clinic day finishing documentation is at elevated burnout risk and is more likely to leave the profession or reduce clinical hours.
Named products include Abridge, Nuance DAX (Dragon Ambient eXperience), Suki AI, and Augmedix. Leading ambient AI scribes report note acceptance rates above 80% with minimal physician editing in clinical pilot deployments, and JAMA studies have demonstrated measurable reductions in after-hours documentation time among physicians using ambient AI compared to traditional documentation workflows. The pattern parallels the evolution of AI-assisted dictation in radiology — a category covered in detail in the structured radiology reporting guide, where AI-assisted dictation and structured reporting are the imaging-specific equivalents of the ambient scribe model used in primary care telemedicine.
5. AI-Assisted Teleradiology and Medical Imaging in Telemedicine
The most technically distinct AI application in telemedicine is the use of AI on medical imaging studies that are acquired at a remote site and interpreted by a radiologist at a different location. Teleradiology — the telemedicine delivery model for radiology — combines with AI in radiology — the diagnostic AI layer — to create a workflow in which the originating site, the AI processing, and the interpreting radiologist may be in three different cities or time zones.
Triage AI tools including Viz.ai for large vessel occlusion stroke detection, Aidoc for multi-pathology triage covering intracranial haemorrhage and pulmonary embolism, and RapidAI for stroke and PE workflows operate effectively in teleradiology contexts where the radiologist is geographically remote from the patient — the AI runs in the cloud, processes the DICOM study at acquisition, and reorders the worklist of a radiologist who may be 1,000 miles from the originating hospital.
AI segmentation supports remote subspecialty consultation: a cardiac MRI acquired at a community hospital can be analyzed with AI ventricular segmentation, with the quantitative measurements delivered to a remote cardiac imaging subspecialist for interpretation. AI-assisted reporting — covered in depth in the AI in radiology guide — integrates with cloud PACS to deliver telemedicine radiology at scale.
The infrastructure layer that enables AI-assisted teleradiology includes a cloud-native PACS with DICOM Gateway acquisition, archive on enterprise cloud infrastructure, AI processing integration via DICOMweb or vendor APIs, a zero-footprint browser-based DICOM viewer for the remote radiologist, and structured reporting with HL7 ORU delivery to the ordering physician’s EHR. The Medicai teleradiology solution provides this complete infrastructure stack.
What AI Can Do in Telemedicine: Measurable Benchmarks
Vendor marketing claims about AI telemedicine performance are abundant. Peer-reviewed clinical validation data are more limited but more useful. The benchmarks below come from published clinical studies and FDA-cleared product validation data.
AI scribe documentation time reduction
JAMA and JAMIA studies on ambient AI scribe deployment have documented reductions in after-hours documentation time of 25% to 40% among physicians using ambient AI compared to traditional documentation workflows, with note acceptance rates above 80% after the initial calibration period. The clinical impact is most pronounced in primary care telemedicine, where documentation burden is highest relative to visit complexity.
AI symptom triage safety advice accuracy
The 2024 BMJ analysis of leading AI symptom checkers found safety advice accuracy ranging from 33% to 79% across products, with significant variability in recommendations for identical symptom presentations. This is the benchmark that should temper enthusiasm: AI triage is useful for routing operational efficiency but is not yet reliable enough to replace clinician judgment for safety-critical triage decisions.
AI ECG arrhythmia detection
The Apple Heart Study published in NEJM enrolled 419,297 participants and demonstrated that the AI-based irregular pulse notification feature successfully identified atrial fibrillation with positive predictive value of 84% in the subgroup that completed confirmatory ECG patch monitoring. FDA-cleared and now deployed at the population scale through consumer wearable devices.
AI-assisted stroke triage in teleradiology workflows
Viz.ai reduced door-to-treatment time for large vessel occlusion stroke by an average of 26 minutes in multi-center deployment studies. In telemedicine stroke care contexts where the interpreting radiologist or stroke neurologist is remote from the patient, the triage AI compresses the radiologist notification interval, which traditionally adds significant delay to remote stroke care.
AI-enhanced telemedicine for resource optimization
A 2025 Nature Scientific Reports analysis found that AI-enhanced telemedicine workflows produced measurable reductions in patient wait times and clinician administrative time across the deployments studied, with the magnitude of improvement dependent on the specific AI tools deployed and the baseline workflow characteristics.
Limitations and Regulatory Status
The capability benchmarks above are real and clinically validated. The limitations that follow are equally real and frequently understated in vendor marketing.
Current Limitations
Variable accuracy across patient populations. AI tools trained predominantly on data from one demographic, geographic, or clinical context underperform when deployed in different contexts. AI symptom checkers calibrated on Western healthcare-seeking populations may underperform in populations with different baseline disease prevalence, presentation patterns, or healthcare engagement norms. The generalisability gap must be evaluated for the specific deployment population, rather than inferred from the original validation study.
Integration friction with EHR and telemedicine platforms. An ambient AI scribe that performs well in a controlled pilot may underperform when integrated into a busy clinic’s EHR workflow because the note format doesn’t match the institution’s documentation standards, the integration adds latency to the visit close-out, or the structured output requires manual reformatting before EHR ingestion. The performance gap between pilot validation and production deployment is consistently underestimated.
Hallucination risk in generative AI. LLM-powered telemedicine tools used for documentation, decision support, or patient-facing communication can produce plausible-sounding but clinically incorrect statements. The risk is highest in tools that generate clinical content (impressions, recommendations, patient-facing summaries) and lower in tools that retrieve and structure existing content. Every AI-generated clinical content requires clinician review and approval — the human-in-the-loop is not optional for generative AI in clinical contexts.
Data privacy and HIPAA considerations. AI processing of telemedicine data — video, audio, clinical text, imaging — creates HIPAA Business Associate relationships with every AI vendor in the data flow. The technical safeguards required for AI processing (encryption, access controls, audit logging) apply with the same force as for any other PHI processing, and the contractual structure (signed Business Associate Agreements with every vendor that touches the data) must be in place before any clinical deployment.
Regulatory Status
The FDA AI/ML-Enabled Medical Devices database includes AI tools used in telemedicine that meet the Software as a Medical Device threshold — primarily diagnostic AI tools used in teleradiology, AI arrhythmia-detection algorithms in wearable cardiac monitors, and AI symptom checkers that claim diagnostic intent. Many AI tools used in telemedicine — ambient scribes that produce clinical documentation rather than diagnostic conclusions, AI clinical decision support that surfaces information without making the decision — fall outside the FDA SaMD definition and are regulated through other pathways.
State-level telemedicine regulation overlays on the federal AI framework. The clinical service delivered through AI-enhanced telemedicine must comply with the medical practice standards of the state where the patient is located — including the requirement that the prescribing provider be licensed in the patient’s state, regardless of where the AI tool was developed or where the provider is physically located. The Interstate Medical Licensure Compact streamlines multi-state licensing but does not eliminate the state-by-state requirement.
HIPAA Business Associate Agreement requirements apply to every AI vendor in the data flow. An AI scribe vendor that processes audio and produces clinical notes is a Business Associate. A symptom triage AI receiving PHI is a Business Associate. The contractual structure required by 45 CFR § 164.504(e) applies regardless of how novel the AI technology is—and the absence of a signed BAA exposes both the healthcare organization and the AI vendor to regulatory and contractual risks.
AI in Telemedicine Compared: Five Application Types at a Glance
| Application type | What it does | Workflow position | Named products | Clinical maturity and validation |
|---|---|---|---|---|
| Virtual visit clinical decision support | Surfaces clinical guidelines, drug interaction alerts, differential diagnosis suggestions, and patient history considerations to the clinician during live video consultations | During the visit — operates alongside the clinician as a real-time reasoning support layer | Epic clinical decision support, Cerner equivalents, Amwell Converge, emerging LLM-powered clinical assistants | Mature — underlying decision support logic validated over two decades of EHR deployment; AI enhancement layer in active expansion as LLMs mature for clinical context integration |
| AI triage and symptom assessment | Collects symptoms before the visit, applies clinical decision algorithms, and routes patients to urgent care, primary care, specialist consultation, or emergency department | Before the visit — operates as a pre-clinical gateway directing demand to the appropriate level of care | Buoy Health, K Health, Ada Health, Babylon Health | Emerging — BMJ 2024 analysis found safety advice accuracy of 33% to 79% across leading products; clinically useful for operational routing but insufficient for safety-critical triage replacement |
| Remote patient monitoring with AI analytics | Processes continuous physiological data from wearables and home medical devices, detecting clinically significant patterns and alerting clinicians when intervention is warranted | Continuously between visits — operates 24/7 on data streams and surfaces actionable alerts to the care team | Apple Watch AFib detection (FDA-cleared), Dexcom CGM with AI pattern analytics, AI-enhanced pulmonary function monitors | Mature for specific applications — Apple Heart Study (NEJM, 419,297 participants) demonstrated AI AFib detection at 84% positive predictive value; CMS RPM codes 99453, 99454, 99457, and 99458 reimburse the monitoring service |
| Ambient AI scribes and clinical documentation | Listens to the consultation, transcribes the conversation, structures it into clinical documentation, and populates the EHR encounter note for clinician review and signature | During and immediately after the visit — produces draft documentation that the clinician reviews, edits where needed, and signs | Abridge, Nuance DAX (Dragon Ambient eXperience), Suki AI, Augmedix | Maturing rapidly — leading products report 80%+ note acceptance rates with minimal clinician editing; JAMA studies document 25–40% reductions in after-hours documentation time |
| AI-assisted teleradiology and medical imaging | Applies CAD, triage prioritisation, segmentation, and reporting AI to medical imaging studies acquired at a remote site and interpreted by a radiologist at a different location | Throughout the imaging workflow — from study acquisition at the originating site through AI processing through remote interpretation through structured report delivery to the ordering physician | Viz.ai (stroke triage), Aidoc (multi-pathology triage), Lunit INSIGHT CXR (chest X-ray), iCAD ProFound AI (mammography), integrated through cloud PACS infrastructure | Mature for triage and CAD — Viz.ai reduced door-to-treatment time for stroke by 26 minutes in multi-centre studies; iCAD ProFound AI demonstrated a 9.6% cancer detection rate increase in mammography validation |
The Intersection of AI, Telemedicine, and Medical Imaging
Why Medical Imaging Is a Distinct AI in Telemedicine Use Case
Most coverage of AI in telemedicine focuses on text and video-based interactions: virtual consultations, AI chatbots, ambient scribes, symptom checkers. The imaging dimension of telemedicine is structurally different and is routinely omitted from generalist analyses. Medical imaging in telemedicine involves data that is large (a typical CT study is 50–200 MB), structured (DICOM format with extensive technical metadata), and that requires specialized software and specialist clinical interpretation. The AI applications that work well for virtual visits — chatbots, ambient scribes, real-time decision support — do not address the imaging dimension. The AI applications that work for imaging — CAD detection, triage prioritization, segmentation, RAG-enhanced reporting — require teleradiology infrastructure to operate at telemedicine scale.
The practical implication is that an organization deploying AI-enhanced telemedicine for primary care does not thereby have AI-enhanced telemedicine for radiology. The two require different tools, infrastructure, and integration architectures. Treating them as a single category — as many vendors do — leads to procurement decisions that satisfy one need at the expense of the other.
How Cloud PACS Enables AI-Assisted Telemedicine for Radiology
The technical architecture for AI-assisted teleradiology as a telemedicine service has four components.
- First, a DICOM Gateway at the originating site forwards the imaging study from the modality (CT, MRI, X-ray, ultrasound) to the cloud archive over a standard HTTPS connection — no VPN, no on-site PACS server required.
- Second, the cloud PACS archive stores DICOM studies with full metadata preservation and makes them accessible to authorized users.
- Third, the AI processing layer — whether built into the PACS platform or integrated via DICOMweb APIs — analyses the study, generates outputs (CAD detections, triage prioritization, segmentation overlays, draft reports), and attaches them to the study in the archive.
- Fourth, the remote radiologist accesses the study via a zero-footprint browser-based DICOM viewer, reviews the AI output alongside the original images, and produces a signed, structured report that is delivered to the ordering physician via HL7 ORU integration with the EHR.
Each of the four components must operate together for the workflow to function. A cloud PACS without AI integration cannot support the AI layer. AI tools without a cloud archive cannot access the DICOM data they need to analyze. A zero-footprint viewer is essential because the remote radiologist may be reading from any device, anywhere. Medicai’s cloud-native PACS on Microsoft Azure provides all four components as a single integrated platform. This infrastructure layer connects AI processing to remote radiologists to ordering physicians through a HIPAA-compliant, audit-logged data flow.
What Practices Need to Deploy AI in Telemedicine Radiology
For a healthcare organization evaluating AI-enhanced teleradiology as a telemedicine service, the practical checklist is short and specific. Cloud PACS with DICOM Gateway support, eliminating the need for on-premise PACS servers at the originating site. A zero-footprint browser-based DICOM viewer with multiplanar reconstruction capability, supporting remote reading from any device. HL7 ORU integration to the ordering physician’s EHR for structured report delivery and FHIR-based image access for clinical context integration. A signed HIPAA Business Associate Agreement from every vendor in the data flow — the PACS vendor, the AI tool vendors, and any intermediate integration platform. AI tool integration through DICOMweb (WADO-RS, QIDO-RS, STOW-RS) for cloud-native tools or vendor-specific API integration for legacy tools.
The Medicai platform — cloud PACS, zero-footprint viewer, AI integration architecture, HIPAA BAA as standard contract term, and HL7/FHIR integration support — meets this complete checklist as a single subscription. See the teleradiology solution page for the full technical architecture and the cloud PACS product page for the underlying infrastructure capabilities.
Frequently Asked Questions
What is AI in telemedicine?
AI in telemedicine is the application of machine learning, natural language processing, and computer vision to support remote healthcare delivery. The scope includes clinical decision support during virtual visits, AI-driven triage and symptom assessment before visits, AI analytics on continuous remote patient monitoring data, ambient AI scribes that handle clinical documentation, and AI-assisted interpretation of medical imaging in teleradiology workflows. AI augments rather than replaces the clinician in telemedicine contexts — every clinical decision and every patient-facing output requires clinician review and approval.
How is AI used in telehealth?
AI in telehealth spans the same five application categories as AI in telemedicine — clinical decision support, triage and symptom assessment, remote patient monitoring with AI analytics, ambient AI scribes, and AI-assisted teleradiology. Still, the telehealth category is broader than telemedicine and includes non-clinical applications such as workforce training, administrative coordination, and public health surveillance. For the regulatory and definitional distinction between the two terms, see the telehealth vs telemedicine guide. In practice, AI applications and underlying technologies are largely the same — the distinction matters mainly for reimbursement and regulatory classification, rather than for the AI technology itself.
What are the benefits of AI in telemedicine?
The primary benefits with cited evidence include reduced after-hours documentation time for clinicians (25–40% reduction with ambient AI scribes in JAMA studies), reduced door-to-treatment time for critical findings in teleradiology workflows (26-minute reduction in stroke care with Viz.ai), continuous physiological monitoring at scale that would be impossible without AI analytics (Apple Heart Study demonstrated AFib detection in 419,000+ participants), operational efficiency in patient routing and triage that reduces low-acuity demand on clinician time, and improved access to specialist consultation for patients in geographies where local specialists are unavailable. The benefits are most measurable in specific application categories — fewer in number than vendor marketing suggests, but real and clinically validated where they exist.
What are the limitations of AI in telemedicine?
The primary limitations include variable accuracy across patient populations not represented in training data, integration friction between AI tools and existing EHR and telemedicine platforms, hallucination risk in generative AI tools that produce clinical content, data privacy and HIPAA Business Associate complexity when multiple AI vendors process PHI, and the underdocumented per-organization financial ROI of many AI tools at the deployment level. Every AI tool in clinical telemedicine requires clinician review and approval of its outputs — the human-in-the-loop is not optional for clinical AI applications, regardless of the AI’s validation performance.
Is AI in telemedicine HIPAA compliant?
AI processing of telemedicine data — video, audio, clinical text, imaging — creates Business Associate relationships under HIPAA with every AI vendor in the data flow. HIPAA compliance is achievable but requires specific contractual and technical infrastructure: signed Business Associate Agreements (45 CFR §164.504(e)) with every vendor, AES-256 encryption of data at rest, TLS 1.2 or higher for data in transit, role-based access controls, complete audit logging of access events, and clear breach notification protocols. The technical safeguards are well-established. The contractual structure must be in place before any clinical deployment — and the absence of a signed BAA with any AI vendor processing PHI exposes both the healthcare organization and the AI vendor to regulatory enforcement and contractual liability.
How does AI work in teleradiology?
AI in teleradiology operates as a layer within the cloud PACS workflow. The DICOM study is forwarded from the originating modality through a DICOM Gateway to the cloud archive, AI tools (Viz.ai for stroke triage, Aidoc for multi-pathology detection, Lunit INSIGHT CXR for chest X-ray, iCAD ProFound AI for mammography, and others) process the study and generate outputs — detections, triage prioritisation, segmentation overlays, or draft report content — that are attached to the study or delivered through separate alerting workflows, and the remote radiologist accesses the study through a zero-footprint browser-based viewer with the AI outputs available alongside the original images. For the comprehensive treatment of AI in radiology, including CAD, triage, segmentation, reporting assistance, and workflow automation, see the AI in radiology guide.
The Future of AI in Telemedicine
Three directions of development are visible in current research and product roadmaps. First, ambient AI maturation continues beyond documentation into clinical reasoning support — AI tools that listen to the consultation and surface relevant clinical considerations to the provider in real time, rather than producing documentation only after the visit ends. Second, multimodal AI models that combine vision, text, and audio for richer virtual consultations — including the ability to incorporate medical images viewed during the consultation into the AI’s contextual reasoning rather than treating imaging as a separate workflow. Third, AI-assisted remote subspecialty consultation expanding from radiology into dermatology, ophthalmology, pathology, and cardiology — applications where image-based or signal-based remote consultation benefits from the same AI-on-cloud-archive architecture that teleradiology has developed.
The regulatory pathway is becoming clearer as the FDA’s Predetermined Change Control Plan framework for AI/ML SaMD matures, enabling iterative model updates without requiring new clearance for each version. State telemedicine regulation continues to evolve toward greater multi-state portability through the Interstate Medical Licensure Compact. CMS coverage policy for AI-enhanced telemedicine services is expanding incrementally, with most current reimbursement directed at the underlying clinical service rather than the AI layer specifically.
For healthcare organisations evaluating where to invest in AI-enhanced telemedicine today, the practical sequence is to start with the application categories that have the strongest evidence base and the clearest reimbursement pathway — remote patient monitoring with AI analytics under CMS RPM codes, AI-assisted teleradiology for triage and CAD where FDA-cleared products are available, ambient AI scribes where physician burnout is a documented organisational problem — and to treat the generative and assistive AI categories as longer-horizon investments where the evidence base and regulatory clarity will continue to develop over the next several years.
Related Articles



Lets get in touch!
Learn more about how Medicai can help you strengthen your practice and improve your patients’ experience. Ready to start your Journey?
Book A Free Demo