CBCT Imaging in Orthodontics: How AI Uses DICOM Data to Plan Treatment Before a Bracket Is Placed
When an orthodontist takes a CBCT scan — a Cone Beam Computed Tomography study that captures the patient’s dental anatomy in three dimensions — the result is a DICOM file. Not a photograph, not a proprietary format specific to the orthodontic software — a DICOM study, the same standardized medical imaging format used in hospital radiology departments to store CT, MRI, and X-ray studies. This is the technical fact that connects modern orthodontic imaging to the broader world of medical imaging infrastructure, and it is the fact that makes AI-assisted orthodontic treatment planning possible at the scale it is beginning to reach.
The AI BRACES™ by SMILE-FX® workflow — where CBCT imaging feeds an AI system that maps the full course of tooth movement before treatment begins — depends entirely on the quality, completeness, and accessibility of the DICOM data the CBCT scanner produces. Understanding what that data contains, how it flows through an orthodontic practice, and what infrastructure it requires is the foundation for understanding why AI is changing orthodontic treatment planning — and why the imaging pipeline matters as much as the AI algorithm itself.
This guide covers the complete picture: what CBCT imaging captures and what DICOM data it produces, how AI uses that data to simulate treatment before a bracket is placed, what storage and sharing infrastructure dental practices need, and why the imaging pipeline determines whether AI-assisted planning delivers on its clinical promise.
What Is CBCT Imaging and What DICOM Data Does It Produce?
Cone Beam Computed Tomography is a specialised form of CT imaging designed for dental and maxillofacial structures. Unlike medical CT, which uses a fan-shaped beam and reconstructs cross-sectional slices from multiple detector positions, CBCT uses a cone-shaped beam that rotates around the patient’s head in a single pass — capturing the full dental anatomy, teeth, roots, alveolar bone, temporomandibular joints, and airway, in one acquisition. The radiation dose is significantly lower than medical CT, and the spatial resolution — submillimeter in modern systems — is sufficient to visualise individual root morphology, bone density variation, and the three-dimensional position of unerupted or impacted teeth.
The output of a CBCT scan is a DICOM dataset — hundreds to thousands of individual DICOM image files, each representing one cross-sectional slice of the acquired volume, each containing both the image pixel data and a DICOM header. The DICOM header encodes the patient identity, the acquisition parameters, the slice geometry, and the three-dimensional spatial coordinates that describe exactly where in the patient’s anatomy each slice was acquired. It is the spatial coordinate data in the DICOM header — the Image Position Patient and Image Orientation Patient tags in the DICOM standard — that enables software to reconstruct the individual 2D slices into a coherent three-dimensional volume: a digital model of the patient’s dentition that can be rotated, measured, and analysed in any plane, from any ang ji le, without additional scanning.
A typical dental CBCT study produces between 300 and 1,500 DICOM files, with a total dataset size ranging from 50 MB to 500 MB depending on the field of view and the resolution settings used. This is the primary dataset that AI treatment planning systems consume — and it is why the quality of the CBCT acquisition directly determines the quality of the AI output that follows.
Why DICOM quality determines AI planning accuracy. AI treatment planning tools for orthodontics are trained on thousands of annotated CBCT cases — labelled datasets where expert orthodontists have identified anatomical landmarks, measured spatial relationships, and classified treatment challenges. These tools perform best when the input DICOM data closely resembles the training data in acquisition quality. Three factors in the CBCT acquisition determine whether the AI output is clinically reliable.
Field of view calibration: the physical dimensions encoded in the DICOM header must accurately reflect the patient’s anatomy for measurements to be correct. A miscalibrated CBCT scanner that reports voxel dimensions inaccurately will cause the AI’s spatial measurements to be systematically wrong — a 1% error in voxel dimension produces a 1% error in every linear measurement across the entire volume. Image noise and artefact levels: metallic restorations, orthodontic brackets, and implants produce beam hardening artefacts — bright streaks and dark bands — that can obscure root morphology in the CBCT volume and cause automated segmentation algorithms to fail or produce inaccurate tooth boundaries. Patient positioning consistency: the patient’s head position during acquisition affects the spatial orientation of the DICOM volume and the accuracy of automated landmark detection. A standardised head positioning protocol — reproducible across all CBCT acquisitions at a practice — ensures that the AI’s reference frame is consistent between the baseline study and any serial studies taken during treatment.
The Reactive vs Predictive Problem in Orthodontics
Traditional orthodontic treatment follows a reactive model. Brackets are placed, the clinician observes how teeth respond over weeks and months, and adjustments are made based on what is observed. This model has worked for decades and is grounded in genuine clinical expertise. But it has a structural limitation: complications are discovered mid-treatment rather than anticipated before treatment begins. Mid-course corrections are common. Treatment timelines extend. Patients told to expect 18 months find themselves still in treatment at 24 or 30 — not because the clinician is unskilled, but because the planning process depends on observation rather than prediction.
Predictive treatment planning addresses this at the source. Rather than responding to how teeth move, the clinician designs the full movement sequence upfront using a digital model of the patient’s dentition — identifying complications before they become clinical problems and designing around them in the simulation rather than correcting them mid-treatment. The technology that makes this possible is the same technology that produces the DICOM dataset described above: CBCT imaging providing the three-dimensional input that AI planning systems require to simulate treatment accurately.
Conventional orthodontic treatment spans 12 to 36 months. Practices applying AI-assisted predictive planning have reported treatment times as short as 4 to 6 months for qualifying cases — not by compressing the biology of tooth movement, which cannot be accelerated beyond physiological limits, but by eliminating the inefficiencies that accumulate in a reactive planning model: the adjustment visits that correct a problem that was not anticipated, the treatment extensions that follow a mid-course complication, the additional appointments generated by a plan that did not account for a root proximity constraint that was present in the CBCT data from day one.
How AI Uses CBCT DICOM Data for Orthodontic Treatment Planning
The AI-assisted orthodontic treatment planning workflow follows a defined sequence from imaging to approved clinical plan. Each step in the sequence is technically dependent on the DICOM data pipeline that precedes it.
Step 1 — CBCT acquisition and DICOM transmission. The patient is positioned in the CBCT scanner and a single-pass acquisition captures the full dental anatomy. The resulting DICOM study is sent from the scanner to the practice’s imaging storage system — either a local PACS server or a cloud PACS archive — where it is stored as a structured DICOM dataset preserving all header metadata.
Step 2 — DICOM import and automated segmentation. The DICOM study is imported into the AI treatment planning software. The software reads the DICOM volume — all slices, all header data — and performs automated segmentation: identifying and separating individual teeth, roots, and alveolar bone structures from the surrounding tissue. This segmentation step is where AI performs its most significant work in the current generation of planning tools. Manual tooth segmentation of a CBCT volume by a trained dental technician previously required 2 to 4 hours of skilled editing. AI-assisted segmentation trained on thousands of annotated CBCT cases now completes the same task in minutes, with accuracy sufficient for clinical use in the majority of cases.
Step 3 — Landmark detection and three-dimensional spatial analysis. The AI system identifies anatomical landmarks across the segmented volume — the midline, the occlusal plane, the position of each tooth’s long axis relative to the alveolar bone, the proximity of roots to critical structures including the inferior alveolar nerve and the cortical bone boundaries. It measures the three-dimensional relationships that define the treatment challenge: crowding, spacing, rotations, tip and torque discrepancies, root proximity to adjacent teeth and bone boundaries, and skeletal discrepancies between the maxilla and mandible. These measurements are extracted directly from the DICOM spatial coordinate data — they are only as accurate as the DICOM header data is complete and correctly populated.
Step 4 — Movement simulation and complication detection. The AI generates a simulated movement sequence — a digital model of the full treatment plan — that maps each tooth’s required movement from its current three-dimensional position to its target position, stepwise through the treatment timeline. At each simulated step, the AI checks for root proximity violations (roots moving too close to each other or to the cortical bone boundary), bone boundary breaches, bite interference between upper and lower arch movements, and alignment with the planned occlusal outcome. Complications identified at this stage are resolved in the simulation before treatment begins. A root proximity constraint that would have appeared as a clinical problem at month six in a reactive planning model appears in the simulation at step four and is designed around before a bracket is placed.
Step 5 — Clinician review, modification, and approval. The orthodontist reviews the complete movement simulation, modifies the sequence where clinical judgment requires it — the AI’s output is a recommendation, not a prescription — and approves the plan. The patient receives a visual projection of their anticipated tooth positions at month three, month six, and at treatment completion. The transparency this provides is a clinical communication tool as much as a technical one: patients who understand where they are going in treatment are better informed partners in the process.
What Dental Practices Need — DICOM Storage, Viewing, and Sharing Infrastructure
The CBCT-to-AI treatment planning workflow creates a DICOM data management requirement that many dental and orthodontic practices are not prepared for at the infrastructure level. The same infrastructure that hospital radiology developed over decades to manage CT, MRI, and X-ray DICOM archives is now required in dental practices managing CBCT studies — and the requirements are technically identical because the data format is technically identical.
Storage: why on-premise servers are becoming impractical. A single CBCT study produces 300 to 1,500 DICOM files with a total study size of 50 MB to 500 MB. A practice performing 10 CBCT scans per week generates 500 MB to 5 GB of new DICOM data weekly — 25 to 260 GB per year. Over five years of active practice, the cumulative CBCT archive reaches 125 GB to 1.3 TB. Unlike 2D dental X-rays, CBCT data cannot be stored in standard dental software proprietary formats without conversion that may strip DICOM header metadata — the same metadata that AI planning tools require for spatial analysis. The data must remain in structured DICOM format to preserve its clinical and computational utility.
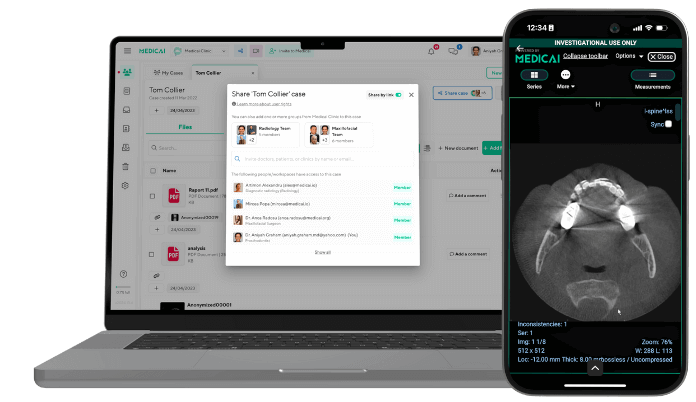
On-premise PACS servers — local server hardware dedicated to DICOM storage — were the standard solution in hospital radiology for decades before cloud infrastructure became reliable enough for clinical data. The same transition from on-premise to cloud is now available to dental practices. Cloud PACS storage on enterprise-grade infrastructure — Microsoft Azure, Amazon Web Services — provides DICOM-compliant object storage without the capital cost of server hardware, without the maintenance burden of on-site IT infrastructure, and with geographic redundancy that local servers cannot match. Medicai’s cloud PACS, built on Microsoft Azure, provides dental and orthodontic practices with the same DICOM archive infrastructure used by hospital radiology departments — subscription-based, no on-premise server required, accessible from any device.
Viewing: why standard dental software is insufficient for CBCT. CBCT DICOM volumes require a viewer that supports multiplanar reconstruction — the ability to reformat the axial acquisition slices into coronal, sagittal, and oblique cross-sectional views, and to generate curved panoramic reconstructions that simulate the traditional dental panoramic X-ray from the CBCT volume. Standard 2D dental imaging software does not support MPR. Dedicated dental CBCT software — Carestream Dental, Planmeca Romexis, Sirona Sidexis — supports these tools but typically requires installation on specific workstations and does not provide browser-based access for reviewing studies from multiple locations or devices.
A zero-footprint browser-based DICOM viewer that supports MPR — the same viewer architecture used in teleradiology and multi-site hospital radiology — provides the CBCT viewing workflow without software installation, without workstation-specific licensing, and without the compatibility problems that arise when different team members use different software versions. The radiologist reviewing a brain MRI remotely from a browser viewer and the orthodontist reviewing a CBCT study remotely from a browser viewer are using the same technical infrastructure for the same clinical purpose: accessing structured DICOM data through a standards-compliant viewer that preserves the spatial information the clinical assessment requires.
Sharing: the CBCT communication challenge. Orthodontic practices regularly share CBCT studies with referring general dentists who ordered the diagnostic assessment, with oral surgeons planning extractions before orthodontic treatment, with periodontists assessing bone levels before or during treatment, and with the patient for informed consent and treatment engagement purposes. A CBCT study of 200 MB cannot be shared by email attachment. A CD or USB drive physically mailed to a referring dentist is slow, creates chain of custody documentation challenges, and cannot be opened by a recipient who does not have compatible CBCT viewing software installed.
Secure link-based DICOM sharing — generating a time-limited encrypted link that opens the full CBCT study in a browser-based zero-footprint viewer, accessible to any recipient from any device without software installation — solves all three problems simultaneously. It is the same mechanism used in teleradiology to share medical imaging studies with remote specialists: the study remains in the cloud archive, the link provides temporary authenticated access to a browser-based view, and the DICOM data never leaves the secure storage environment as an unencrypted file. For a referring dentist receiving a CBCT study from an orthodontic practice, opening a browser link is faster, simpler, and more clinically useful than receiving and opening a CD.
Why AI Changes What the Imaging Infrastructure Must Do
The shift from reactive orthodontics to AI-assisted predictive treatment planning changes the imaging infrastructure requirement in one specific and important way: the CBCT study is no longer just a diagnostic image to be viewed and filed — it is the primary data input for a computational process that must be repeatable, comparable, and auditable across the full course of treatment.
Lossless storage is non-negotiable. The AI planning system must access the original CBCT DICOM study at any point during treatment — to compare the current clinical situation against the planned simulation, to recalculate the remaining movement sequence if a mid-course modification is needed, or to document the final outcome against the original plan for clinical audit and research. A DICOM archive that applies lossy compression during storage — reducing file size by degrading image quality, a default setting in some cloud storage services — corrupts the spatial precision that the AI system requires for accurate comparison and recalculation. Voxel dimension accuracy matters for AI spatial measurement at the level of tenths of millimetres. Lossy compression introduces reconstruction artefacts at exactly the scale that orthodontic measurements are made. Lossless DICOM storage — using DICOM transfer syntaxes that apply only lossless compression or no compression — is the non-negotiable technical baseline for any CBCT archive that feeds an AI treatment planning workflow.
Serial study comparability requires full DICOM metadata preservation. Monitoring treatment progress with AI tools increasingly involves acquiring serial CBCT studies at defined intervals — typically at the midpoint and completion of treatment — to assess how actual tooth movement tracks against the planned simulation. These serial studies must be stored alongside the baseline CBCT in a format that enables automated co-registration: aligning the serial study to the baseline geometry using the DICOM spatial coordinate metadata so that the AI can compare actual vs planned tooth positions at each time point without manual alignment. A cloud PACS that stores each study with complete DICOM header metadata — including Image Position Patient, Image Orientation Patient, Pixel Spacing, and Slice Thickness tags — enables this automated co-registration. A storage platform that strips or modifies DICOM header data during ingestion does not.
Audit logging is becoming a clinical and regulatory requirement. As AI-generated treatment plans become more prevalent in orthodontics, the requirement to document what the AI recommended, what the clinician approved, and what occurred during treatment will become more explicit in professional liability and regulatory frameworks — following the pattern already established in medical imaging, where radiology AI outputs must be documented as part of the clinical record. The CBCT study that formed the basis of the AI plan, the DICOM-encoded treatment simulation, and the serial CBCT studies documenting treatment progress together constitute the clinical record of an AI-assisted treatment episode. A cloud PACS with complete access audit logging — recording who accessed each study, when, and from which device — provides the access trail that this clinical record requires and that a practice needs to demonstrate if the AI-assisted treatment outcome is ever subject to clinical review.
CBCT and Dental PACS: What the Market Looks Like Now
The DICOM infrastructure requirement described above has given rise to a dental PACS market — cloud-based DICOM storage and viewing platforms designed specifically for dental and orthodontic CBCT workflows. The queries practitioners use to research these platforms — dental pacs, dental cbct, cone beam ct dental — are captured in the SERP by RadiologyInfo.org, the FDA, Wikipedia, and dental school academic resources: definition and regulatory content rather than vendor content. No imaging infrastructure vendor currently ranks prominently for these queries.
The dental PACS market currently divides into three categories. Dedicated dental imaging software platforms — Carestream Dental, Planmeca, Dentsply Sirona, Apteryx — provide CBCT acquisition software with proprietary storage and viewing tools built around the scanner hardware they sell. These platforms provide deep integration with specific scanner models but create proprietary data silos that complicate multi-practice workflows and are difficult to integrate with third-party AI planning tools that expect standard DICOM input. Dedicated dental cloud PACS platforms — a smaller and newer category — provide DICOM-compliant cloud storage and viewing designed for the dental CBCT data format, typically without the scanner hardware dependency of the first category. Medical imaging cloud PACS platforms — including Medicai — provide DICOM infrastructure built for hospital radiology that is equally applicable to dental CBCT storage, viewing, and sharing, because the DICOM standard is the same regardless of whether the study comes from a hospital CT scanner or a dental CBCT unit.
For orthodontic practices evaluating cloud infrastructure for CBCT management, the practical evaluation criteria are identical to those applied in the VNA benefits framework developed for medical imaging: does the platform store DICOM losslessly; does the viewer support MPR; does secure external sharing work without recipient software installation; is HIPAA compliance contractual with a signed BAA; and does the platform support the integration with AI planning tools through a standard DICOM or DICOMweb interface?
The Convergence of Dental and Medical Imaging Infrastructure
The trajectory described in the AI BRACES™ by SMILE-FX® methodology — from reactive adjustment to data-informed predictive planning — is not unique to orthodontics. The same shift is occurring across medical imaging: in oncology, where serial CT staging studies feed AI-assisted RECIST measurement tools; in neurology, where brain MRI series feed AI lesion detection and quantification workflows; in radiology broadly, where AI triage tools process DICOM worklists to prioritise urgent findings and flag incidental findings for follow-up. In every case the common denominator is DICOM data — and the infrastructure requirement is the same: store it without loss, retrieve it at speed, view it with the clinical tools the workflow requires, and share it securely with every stakeholder in the care pathway.
Orthodontics is arriving at this infrastructure requirement later than hospital radiology, but for the same technical reasons. The AI planning system requires structured DICOM input. The structured DICOM input requires a compliant archive. The compliant archive requires the same cloud PACS infrastructure that hospital radiology has been standardising for two decades.
The technology is converging. The data format has always been the same. The infrastructure requirements are now the same. And the practices that invest in building the correct DICOM pipeline — lossless storage, MPR-capable viewing, secure sharing, audit logging — will be the ones whose AI treatment planning tools deliver the clinical accuracy and efficiency those tools are capable of.
Frequently Asked Questions
What is a CBCT scan in dentistry?
A CBCT scan (Cone Beam Computed Tomography) in dentistry is a specialised three-dimensional X-ray that captures the full dental anatomy — teeth, roots, bone, temporomandibular joints, and airway — in a single rotation around the patient’s head. Unlike a standard dental X-ray, which produces a two-dimensional image, CBCT produces a three-dimensional DICOM dataset that can be viewed in any plane and measured in any direction. It is used in orthodontics for treatment planning, in oral surgery for implant planning and impacted tooth assessment, in endodontics for root canal evaluation, and in periodontics for bone level assessment. The radiation dose is significantly lower than a medical CT scan while providing submillimeter spatial resolution sufficient for detailed dental and skeletal assessment.
How does AI use CBCT data for orthodontic treatment planning?
AI orthodontic treatment planning tools use the three-dimensional DICOM volume from a CBCT scan as their primary input. The AI first performs automated segmentation — identifying and separating individual teeth, roots, and bone structures from the surrounding tissue in the DICOM volume. It then detects anatomical landmarks, measures three-dimensional spatial relationships between teeth and bone, and simulates the full treatment movement sequence — mapping each tooth from its current position to its target position while checking for root proximity violations, bone boundary breaches, and bite interference at each simulated step. The output is a digital treatment simulation that the orthodontist reviews, modifies where clinical judgment requires it, and approves before any hardware is placed. The clinical value is that complications detectable in the CBCT data — root proximity constraints, bone boundary limitations, skeletal discrepancies — are identified and designed around in the simulation rather than discovered during treatment.
What is the difference between a dental CBCT scan and a regular dental X-ray?
A regular dental X-ray — bitewing, periapical, or panoramic — produces a two-dimensional projection image that compresses three-dimensional anatomy onto a flat plane. Overlapping structures cannot be separated, depth cannot be measured, and root morphology in three dimensions cannot be assessed. A CBCT scan produces a three-dimensional volume in DICOM format that can be reformatted and measured in any plane. This makes CBCT clinically superior for any assessment that requires three-dimensional information: the exact position of an impacted tooth relative to adjacent roots, the bone volume available for an implant, the root morphology before orthodontic movement, or the three-dimensional skeletal relationship between the upper and lower jaws. The trade-off is radiation dose — CBCT delivers a higher dose than a standard dental X-ray, though significantly less than a medical CT — and cost. CBCT is ordered when the clinical question cannot be adequately answered by a two-dimensional image.
What is a dental PACS and does an orthodontic practice need one?
A dental PACS (Picture Archiving and Communication System) is a platform that stores, manages, and provides access to dental imaging studies — primarily CBCT DICOM datasets — in a structured, standards-compliant archive. An orthodontic practice that performs CBCT scanning needs a PACS or equivalent DICOM archive for three reasons: CBCT datasets are too large to store reliably in standard dental practice management software; the DICOM format and metadata that AI planning tools require must be preserved exactly as acquired; and regulatory requirements in most jurisdictions mandate long-term retention of diagnostic imaging records (typically 7 to 10 years) in a format that remains accessible and auditable. Cloud PACS platforms built on enterprise cloud infrastructure provide orthodontic practices with compliant DICOM storage without on-premise server hardware, with subscription pricing that scales with storage volume rather than requiring a capital investment in server hardware.
How is CBCT DICOM data stored and shared in dental practice?
CBCT DICOM data should be stored in a DICOM-compliant archive — either an on-premise PACS server or a cloud PACS platform — that preserves the full DICOM header metadata and applies lossless compression or no compression during storage. Lossy compression that degrades spatial precision is incompatible with AI treatment planning tools that make measurements at submillimeter accuracy from the DICOM volume. For sharing, the most practical and secure mechanism is link-based DICOM sharing: the CBCT study remains in the cloud archive, and a time-limited encrypted link opens the study in a browser-based zero-footprint DICOM viewer accessible to any recipient — referring dentist, oral surgeon, periodontist, or patient — from any device without software installation. This approach preserves the full DICOM dataset in the secure archive while providing access without the compliance and logistics problems of CD distribution or email attachment of large DICOM files.
Conclusion
The AI BRACES™ by SMILE-FX® workflow begins with a CBCT scanner and ends with a treatment plan that anticipates complications before a bracket is placed. Between those two points is a DICOM pipeline — acquisition, storage, retrieval, viewing, and sharing — that determines whether the AI system receives the data quality it needs to produce a clinically reliable simulation. The orthodontist’s skill and the AI algorithm’s sophistication are both limited by the accuracy and completeness of the DICOM data they work with.
As AI-assisted treatment planning becomes standard practice in orthodontics, the imaging infrastructure that supports it will need to meet the same standards that medical imaging has developed over decades: lossless storage that preserves spatial precision, viewer tools that support three-dimensional assessment, and secure sharing mechanisms that give every stakeholder in the care pathway access to the same data. The technology is converging. The data format has always been the same. The infrastructure requirements are now the same. And the practices that build the correct DICOM pipeline will be the ones whose AI treatment planning tools deliver the clinical outcomes those tools are capable of.
Related Articles



Lets get in touch!
Learn more about how Medicai can help you strengthen your practice and improve your patients’ experience. Ready to start your Journey?
Book A Free Demo