PACS vs MIMPS: What changed, and what should you call the system
PACS vs MIMPS is mostly a naming and scope update; the FDA now uses MIMPS as the regulatory name for software systems that manage and process medical images for clinical interpretation.
PACS is the legacy term most hospitals still use. MIMPS is the current FDA term that better matches how modern PACS platforms support processing, quantification, and interpretation workflows, not just storage and sharing.
This blog addresses MIMPS in the context of medical imaging informatics — specifically, the FDA’s updated regulatory classification for imaging software under 21 CFR 892.2050. MIMPS here stands for Medical Image Management and Processing System: the current regulatory name for the class of software that radiologists and hospital IT teams have traditionally called PACS (Picture Archiving and Communication System).
If you arrived here from a PACS search and are trying to determine whether the two terms refer to the same thing, the comparison table below provides the clearest answer.
What is MIMPS
MIMPS means Medical Image Management and Processing System. MIMPS is defined in 21 CFR 892.2050 as a device that provides one or more capabilities for review and digital processing of medical images for interpretation by a trained practitioner for disease detection, diagnosis, or patient management.
MIMPS classification is Class II with special controls. The regulation lists voluntary standards that commonly include DICOM, JPEG, and SMPTE test patterns.
What is PACS
PACS means Picture Archiving and Communication System. PACS traditionally refers to a system that stores, retrieves, and distributes imaging studies, usually in DICOM format, and provides clinical access through a viewer.
PACS usage in hospitals remains common because “PACS” is the familiar operational word for the imaging archive and viewer stack. PACS usage in vendor marketing remains common because buyers still search and budget under “PACS.”
PACS vs MIMPS in one line
PACS is the legacy industry label for the imaging archive and access system, and MIMPS is the FDA’s current regulatory label that explicitly includes image processing for clinical interpretation.
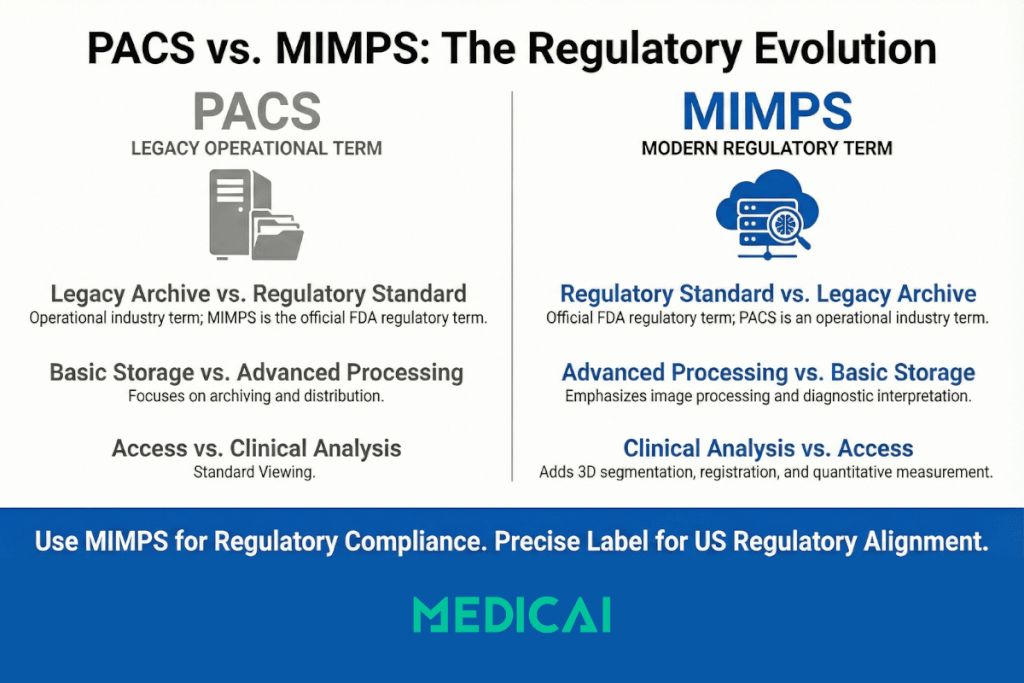
PACS vs MIMPS, practical comparison
The table below compares scope, clinical use, regulated functions, and the words buyers use.
| Aspect | PACS | MIMPS |
| Primary meaning | Legacy industry term for imaging archive, distribution, and viewing | FDA regulatory term for image management and processing for interpretation |
| Core scope | Store, index, retrieve, and distribute studies and priors | Manage images plus digital processing for diagnosis and patient management |
| Processing emphasis | Archiving and communication, basic viewing tools | Processing and interpretation support, advanced manipulation, and quantification |
| Typical capabilities | Archive, search, retrieval, routing, worklists, viewer access, sharing | 3D, segmentation, registration, quantitative measurement, sometimes AI-assisted |
| Clinical workflow role | Access to studies and priors across sites | Access plus analysis and interpretation workflows across specialties |
| Standards layer | DICOM imaging objects and exchange | DICOM plus processing tied to interpretation |
| How people talk | Hospital operations and procurement language | US regulatory and compliance language |
| Safest phrasing | PACS (FDA term, MIMPS) | MIMPS (formerly called PACS) |
| Practical takeaway | Modern PACS and MIMPS often describe the same category | MIMPS is the precise label when regulatory alignment matters |
Why the FDA renamed PACS to MIMPS
The FDA updated the PACS classification name in an April 2021 rulemaking under 21 CFR 892.2050. The amendment history in the CFR points to an April 19, 2021, Federal Register update.
The naming change aligns with a regulatory distinction between simple data handling versus clinical interpretation functions. FDA guidance and commentary on these changes explain that storage, transfer, conversion, and display functions may fall under lower levels of regulatory oversight in specific contexts, while complex image processing for interpretation remains a device function.
MIMPS naming matters because modern “PACS” platforms often include processing features beyond archiving and communication. MIMPS naming makes that expanded capability set explicit in regulation.
What capabilities make a system “MIMPS” in practice
MIMPS capability scope is about interpretation support. MIMPS capability scope usually includes at least 5 categories.
Viewing and diagnostic review
MIMPS supports fast study review for interpretation by trained practitioners across CT, MRI, x-ray, ultrasound, and similar modalities.
Image manipulation for diagnosis
MIMPS supports window and level, zoom, pan, and enhancement functions that influence interpretation quality and speed.
Advanced processing and analysis
MIMPS may include 3D visualization, multimodality registration, segmentation, and quantification workflows, depending on the product and intended use.
Image segmentation in MIMPS
Image segmentation is one of the advanced image manipulation functions explicitly listed in the MIMPS regulatory definition. Segmentation identifies and delineates structures, lesions, or regions of interest within imaging datasets — distinguishing, for example, a hepatic lesion from surrounding liver parenchyma, or the left ventricular myocardium from the blood pool in cardiac MRI.
The clinical contexts where segmentation functions appear most frequently in MIMPS workflows include oncology imaging (lesion detection, volume measurement, and treatment response assessment), radiation therapy planning (target volume delineation), and AI-assisted diagnostic workflows (where segmentation is often the output the AI algorithm returns and the MIMPS platform must display, store, and make auditable).
A MIMPS platform that supports only manual segmentation tools provides a different capability level than one that integrates semi-automated or AI-driven segmentation, and the cleared device’s intended regulatory use should specify which level of segmentation automation is within scope.
Multimodality image registration in MIMPS
Multimodality image registration aligns imaging datasets acquired with different techniques or at different time points, enabling them to be viewed as a spatially coherent composite. Common registration pairs include PET-CT (functional metabolism overlaid on anatomical structure), MRI-PET (soft-tissue anatomy with metabolic activity), and pre- and post-treatment studies within the same modality, where anatomical changes must be tracked against a baseline.
Registration functions that are integrated into the MIMPS reading workflow — rather than handled by a separate processing workstation that exports results back to the archive — are a distinguishing feature of enterprise-grade MIMPS platforms. The clinical significance is that the radiologist or reading clinician can perform or review the registration within the same environment where they complete their interpretation, rather than switching between systems and manually reconciling results.
For procurement teams, the relevant questions are: Is registration performed within the MIMPS viewer or on a separate workstation? Are registered composite datasets stored back to the archive as DICOM objects? Is the registration algorithm’s intended use within the scope of the 510(k) clearance? Each answer affects both clinical utility and regulatory accountability.
Quantitative measurements and tracking
MIMPS often supports measurement tools and time-series comparisons for lesion tracking and follow-up assessment, especially in oncology and cardiology imaging workflows.
Workflow connectivity around images
MIMPS typically integrates with enterprise systems that carry orders, demographics, and results, and it relies on standards such as DICOM in the imaging layer.
What does not change when you move from “PACS” wording to “MIMPS” wording
The day-to-day PACS workflow remains the same for most users. The radiologist still opens a worklist, loads priors, manipulates images, and finalizes interpretation. The clinician still opens the study from the chart or a viewer link.
The main change is aligning the audience and compliance. The term MIMPS signals regulated interpretation and processing functions. The term PACS signals operational imaging infrastructure.
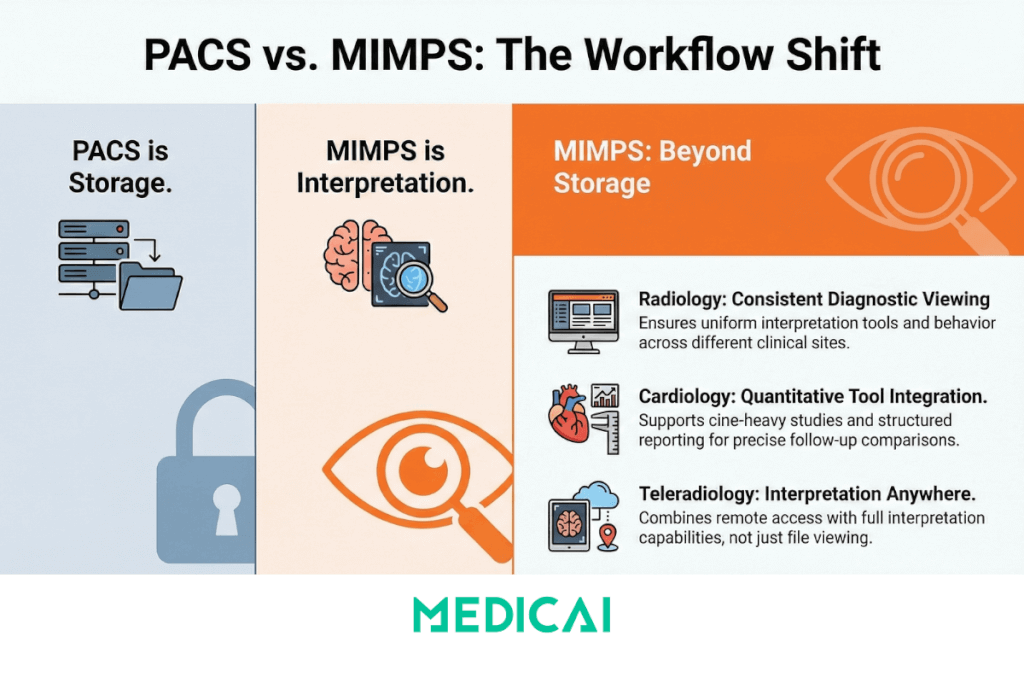
Clinical workflow implications of PACS versus MIMPS
MIMPS language is best suited for describing interpretation-grade imaging workflows. PACS language is best suited for describing archive access and distribution.
Radiology workflow
MIMPS highlights interpretation tooling, processing features, and consistent diagnostic viewing behavior across sites.
Cardiology workflow
MIMPS framing fits cine-heavy studies and quantitative tools that support structured reporting and follow-up comparisons.
Teleradiology and multi-site access
MIMPS framing supports the “reading anywhere” story because it implies both access and interpretation tooling, not only storage.
Procurement and compliance checklist
This checklist definition covers what a buyer or product marketer needs to confirm before using MIMPS language.
Regulatory language alignment
MIMPS phrasing matches the FDA regulation name for 892.2050 in the US context.
Intended use clarity
MIMPS implies interpretation support. PACS phrasing can imply storage and communication only. Your copy must match the product’s intended use and the claims that have been cleared.
Standards alignment
MIMPS systems commonly align with DICOM and related imaging standards referenced in the regulation’s special controls section.
Which voluntary standards does the MIMPS classification reference?
The special controls section of 21 CFR 892.2050 identifies three voluntary consensus standards that are relevant to MIMPS compliance. The regulation’s listing of voluntary standards does not mandate implementing each one in full, but it signals the technical baseline the FDA expects MIMPS platforms to address and that buyers should verify against in conformance documentation.
DICOM
DICOM (Digital Imaging and Communications in Medicine) is the structural interoperability standard for medical imaging. It defines how imaging objects are encoded, identified, stored, retrieved, and exchanged across systems. A MIMPS that fully implements DICOM can interoperate with any standards-compliant modality, archive, viewer, or integration engine. DICOM is the most widely cited standard in MIMPS procurement and the one most likely to appear in conformance statements and 510(k) documentation.
JPEG (ISO/IEC 10918)
JPEG is referenced in the MIMPS special controls for image encoding. In radiology workflows, JPEG encoding applies to image distribution, web viewer delivery (DICOMweb’s WADO-RS uses JPEG among other transfer syntaxes), and integration with non-radiology clinical systems that consume compressed previews. Procurement teams reviewing MIMPS conformance should verify the platform’s supported JPEG transfer syntaxes and document the compression ratio policy for diagnostic versus non-diagnostic use cases. Lossy JPEG compression at aggressive ratios can degrade image quality below diagnostic standards.
SMPTE test patterns
SMPTE (Society of Motion Picture and Television Engineers) test patterns are used to verify that diagnostic display monitors meet the luminance, contrast ratio, and spatial resolution requirements for clinical image interpretation. The SMPTE TG18 test pattern suite is the standard reference for radiology monitor QA programs. A MIMPS deployment that includes high-resolution diagnostic monitors should maintain a documented QA program referencing SMPTE standards to confirm that display conditions remain within the cleared use environment. This matters most for multi-site and teleradiology deployments where monitors vary across locations.
These three standards operate at different layers of the MIMPS technical stack: DICOM governs data structure and transport, JPEG governs image encoding, and SMPTE governs display quality verification. For procurement teams writing conformance requirements, all three should appear in the vendor’s response documentation.
Boundary language
Storage-only and transfer-only functions may fall into different regulatory buckets in some contexts. FDA guidance discusses MDDS and related image storage and communication device categories, so your copy should avoid implying “interpretation” when the product is only for storage or transport.
FAQs
What does MIMPS stand for in radiology?
MIMPS stands for Medical Image Management and Processing System, the FDA classification name for systems that support review and digital processing of medical images for interpretation.
Did the FDA replace PACS with MIMPS?
The FDA updated the regulation title and classification language so the relevant regulation now uses “medical image management and processing system,” with an amendment recorded in 2021.
Is MIMPS different from modern PACS?
MIMPS and modern PACS often refer to the same platform category in practice, but MIMPS explicitly emphasizes processing and interpretation support, while PACS remains the legacy operational term.
When should you say “MIMPS” instead of “PACS”?
MIMPS language fits regulatory, technical, and up-to-date audiences in the US. PACS language fits day-to-day hospital buying and clinical conversation, because PACS remains the term people search and use internally.
Related Articles



Lets get in touch!
Learn more about how Medicai can help you strengthen your practice and improve your patients’ experience. Ready to start your Journey?
Book A Free Demo